Some very disappointing news released today, as seen on Reuters:
Oct 18 (Reuters) - Abbott Laboratories Inc said its partner Reata Pharmaceuticals was discontinuing a late-stage trial of their potential blockbuster treatment for chronic kidney disease and diabetes based on safety concerns raised by an independent safety committee. The news for bardoxolone represents a major setback for Abbott just months before the planned Jan. 1 spinoff of its branded prescription drugs into a separate publicly traded company called AbbVie. Without the high-profile drug, Wall Street concerns about AbbVie's dependence on Abbott's $8 billion-a-year rheumatoid arthritis drug Humira could intensify. An independent data monitoring committee found excess serious adverse events and mortality in patients taking the oral anti-inflammatory drug, Abbott said in a regulatory filing.
Regulators were notified of the decision, and study participants were being informed, the company said.
 3,883 patients with secondary hyperparathyroidism, those on active treatment received a titrated dose of cinacalcet starting at 30 mg/day up to 180mg and were followed for approximately 4 years. Patients were evaluated for the composite primary outcome of all-cause mortality or first non-fatal cardiovascular event, including myocardial infarction, hospitalization for unstable angina, heart failure or peripheral vascular event. Despite very promising biochemical outcomes from earlier studies with cinacalcet, the reduction in the primary outcome, while numerically positive, was not statistically significant. A more detailed analysis of this study will be completed once the paper is published.
3,883 patients with secondary hyperparathyroidism, those on active treatment received a titrated dose of cinacalcet starting at 30 mg/day up to 180mg and were followed for approximately 4 years. Patients were evaluated for the composite primary outcome of all-cause mortality or first non-fatal cardiovascular event, including myocardial infarction, hospitalization for unstable angina, heart failure or peripheral vascular event. Despite very promising biochemical outcomes from earlier studies with cinacalcet, the reduction in the primary outcome, while numerically positive, was not statistically significant. A more detailed analysis of this study will be completed once the paper is published.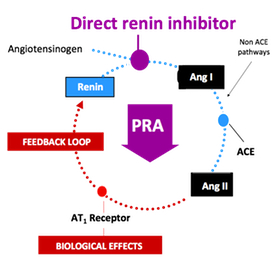 In a stunning development, Novartis said Tuesday that it will terminate the late-stage ALTITUDE study investigating Rasilez (aliskiren) in patients with type 2 diabetes and renal impairment on the recommendation of an independent data monitoring committee. The company indicated that the committee concluded that "patients were unlikely to benefit" from the addition of Rasilez to standard anti-hypertensives and also identified higher adverse events in this group (source:
In a stunning development, Novartis said Tuesday that it will terminate the late-stage ALTITUDE study investigating Rasilez (aliskiren) in patients with type 2 diabetes and renal impairment on the recommendation of an independent data monitoring committee. The company indicated that the committee concluded that "patients were unlikely to benefit" from the addition of Rasilez to standard anti-hypertensives and also identified higher adverse events in this group (source: 